Interflox Oral
Interchemie,Netherlands
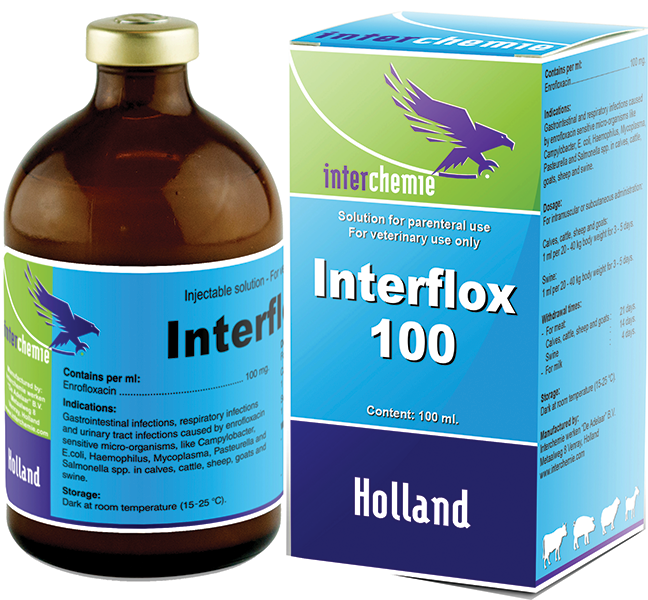
Solution for injection
Product name:
Interflox - 100.
Description:
injection solution. 100 ml.
Composition of each ml:
Enrofloxacin ................................................... ................................................ .......100 mg;
target species
Calf, sheep, goat and pig.
Clinical pharmacology
Pharmaceutical group:
Antibacterial agents of quinolones and quinoxaline, fluoroquinolones.
ATCvet code:
QJ01MA90
Enrofloxacin belongs to the group of quinolines, characterized by antimicrobial activity against such bacteria as Actinobacillus pleuropneumoniae, E.coli, Klebsiella spp., Mannheimia haemolytica, Mycoplasma spp., Pasteurella multocida and Staphylococcus aureus.
Indications:
Cattle
Treatment of respiratory tract infections caused by strains susceptible to enrofloxacin such as Pasteurella multocida, Mannheimia haemolytica and Mycoplasma spp.
Treatment of acute severe mastitis caused by Escherichia coli strains susceptible to enrofloxacin.
Treatment of gastrointestinal tract infections caused by strains of Escherichia coli susceptible to enrofloxacin.
Treatment of sepsis caused by strains of Escherichia coli susceptible to enrofloxacin.
Treatment of acute mycoplasma-associated arthritis caused by enrofloxacin-susceptible strains of Mycoplasma bovis in cattle under 2 years of age.
sheep
Treatment of gastrointestinal tract infections caused by strains of Escherichia coli susceptible to enrofloxacin.
Treatment of sepsis caused by strains of Escherichia coli susceptible to enrofloxacin.
Treatment of mastitis caused by strains of Staphylococcus aureus and Escherichia coli susceptible to enrofloxacin.
a goat
Treatment of respiratory tract infections caused by strains of Pasteurella multocida and Mannheimia haemolytica susceptible to enrofloxacin.
Treatment of gastrointestinal tract infections caused by strains of Escherichia coli susceptible to enrofloxacin.
Treatment of sepsis caused by strains of Escherichia coli susceptible to enrofloxacin.
Treatment of mastitis caused by strains of Staphylococcus aureus and Escherichia coli susceptible to enrofloxacin.
pig
Treatment of respiratory tract infections caused by strains of Pasteurella multocida, Mycoplasma spp and Actinobacillus pleuropneumoniae susceptible to enrofloxacin.
Treatment of urinary tract infections caused by strains of Escherichia coli susceptible to enrofloxacin.
Treatment of post-profit dysgalactic syndrome PDS (MMA syndrome) caused by enrofloxacin-susceptible strains of Escherichia coli and Klebsiella spp.
Treatment of gastrointestinal tract infections caused by strains of Escherichia coli susceptible to enrofloxacin.
Treatment of sepsis caused by strains of Escherichia coli susceptible to enrofloxacin.
Contraindications:
Do not use in known animal hypersensitivity to enrofloxacin, other fluoroquinolones or any of the excipients. Do not use in young horses due to possible articular cartilage damage.
Adverse side effects:
In very rare cases, disturbances in the function of the digestive tract (eg diarrhea) may develop. These symptoms are usually mild and transient. In very rare cases, intravenous treatment of cattle can cause shock reactions, which are probably the result of impaired blood circulation. Local reactions at the injection site. In pigs, after intramuscular administration of the drug, inflammatory reactions may develop, which may persist for approximately 28 days after injection.
Interaction:
The use of enrofloxacin is not allowed with antimicrobial agents that have an antagonistic effect on quinolones (for example, macrolides, tetracyclines or phenicols). Do not use at the same time as theophylline, as this may slow down the elimination of theophylline from the body.
Use during pregnancy and lactation
Cattle: The safety of the veterinary medicinal product has been established in dairy cows during the first trimester. The drug can be used in cows in the first trimester of pregnancy. The use of the product in cows during the last three trimesters of pregnancy should be based on the judgment of the responsible veterinarian regarding the risk-benefit ratio of the use of the medication. The product can be used in cows during lactation.
Sheep and goats: the safety of the veterinary drug during pregnancy and lactation has not been established. Use only in accordance with a risk-benefit assessment of the use of the medication by a responsible veterinarian.
Pigs: The safety of the veterinary medicinal product during pregnancy has not been established. Use only in accordance with a risk-benefit assessment of the use of the medication by a responsible veterinarian. The drug can be used in mother pigs during the lactation period.
Dosage:
Intravenous, subcutaneous or intramuscular use.
Repeated injections should be done at different injection sites.
To ensure the correct dosage, it is necessary to determine the body weight as accurately as possible to avoid the use of an insufficient dose.
Cattle:
5 mg enrofloxacin/kg body weight, equivalent to 1 ml/20 kg body weight, once daily for 3-5 days.
Treatment of acute mycoplasma-associated arthritis in animals less than 2 years of age caused by Mycoplasma Bovis strains susceptible to enrofloxacin: 5 mg enrofloxacin/kg body weight, equivalent to 1 ml/20 kg body weight, once daily for 5 days.
The product is used slowly intravenously or as a subcutaneous injection.
Acute mastitis caused by Escherichia coli: 5 mg enrofloxacin/kg body weight, equivalent to 1 ml/20 kg body weight, once a day and for 2 subsequent days. A second dose may be administered subcutaneously. In this case, the period of exit from the body will be corresponding to subcutaneous administration.
During each subcutaneous injection, no more than 10 ml should be injected into one injection site.
Sheep and goats:
Enrofloxacin 5 mg/kg body weight, which is equivalent to 1 ml/20 kg body weight, once a day as a subcutaneous injection for 3 days.
During each subcutaneous injection, no more than 6 ml should be injected into one injection site.
Pig:
Enrofloxacin 2.5 mg/kg body weight, equivalent to 0.5 ml/20 kg body weight, once daily intramuscularly for 3 days.
For gastrointestinal infection or septicemia caused by Escherichia coli: enrofloxacin 5 mg/kg body weight, equivalent to 1 ml/20 kg body weight, once daily intramuscularly for 3 days.
In pigs, the injection should be made in the neck area at the base of the ear.
During each subcutaneous injection, no more than 3 ml should be injected into one injection site.
Waiting period:
Cattle: after intravenous injection:
Meat and offal: 5 days.
Milk: 3 days.
After subcutaneous injection:
Meat and offal: 12 days.
Milk: 4 days.
Sheep: meat and offal: 4 days.
Milk: 3 days.
Goat: meat and offal: 6 days.
Milk: 4 days.
Pig: meat and offal: 13 days.
Keep out of reach of children!
Storage conditions:
The shelf life of Interflox-100 is 3 years if stored in a dark place at a temperature below 25°C. After first opening, Interflox-100 can be stored for 2 weeks in a refrigerator (2-8 °C) away from light.
Manufacturer: Interchemie werken “De Adelaar” B.V. Metaalweg 8, 5804 CG Venray, The Netherlands
Importer and exclusive distributor in Georgia "Invet LLC" Address: St. Tbilisi, Vakhushti Bagrationi str. 84a, tel.: +995 32 2 25 19 66;